
Lupus and COVID-19 Vaccination – UPDATE
 February 07th, 2024
February 07th, 2024 Nakita Cambow
Nakita Cambow Latest News
Latest News 0 Comments
0 Comments
UPDATED 08/02/2024
In this article we will do our best to provide reliable information and guidance about COVID-19 vaccination for people with lupus. This will be updated regularly over the coming weeks and months as new information becomes available – please check back frequently.
People with lupus often have very different symptoms and treatments from each other and we are unable to give advice on case-by-case basis; some guidance may therefore be very general.
Latest Updates
Eligibility for 2024 booster vaccine
The groups eligible for the 2024 spring booster have been announced. You can read more about it below – READ MORE
Children aged 6 months to 4 years in clinical risk groups will be offered the vaccine from June 2023.
You can read more about it below – READ MORE
Contents
Click on any of the below to be taken to the relevant section:
- How is the COVID-19 vaccine given?
- Should people with lupus have the COVID-19 vaccine?
- Is the COVID-19 vaccine safe for people with lupus?
- Should I have the vaccine if I am experiencing a flare?
- What about people with a weakened immune system and those taking immunosuppressant medication(s)?
- Will I be offered a third vaccine dose?
- Do I need a booster dose?
- Am I eligible for the 2024 booster dose programme?
- Information on vaccinations for children aged 6 months to 4 years.
- What should I do if I am taking anticoagulation therapy?
- Who SHOULD NOT have the COVID-19 vaccine?
- Is the COVID-19 vaccine effective?
- Does the COVID-19 vaccine have side-effects?
- Which COVID-19 vaccine will I be given?
- Further reading
How is the COVID-19 vaccine given?
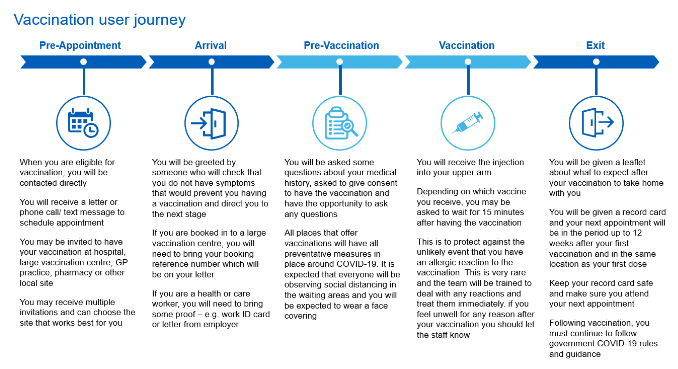
Pre-Appointment
When you are eligible for vaccination, you will be contacted directly. You will receive a letter, phone call or text message to schedule your appointment. You may be invited to have your vaccination at a hospital, a large vaccination centre, a GP practice, a pharmacy or other local site. You may receive multiple invitations – you should choose the option that suits you best.
Arrival & Pre-Vaccination
 Upon arrival at your vaccination centre you will be greeted by someone to check that you do not have symptoms that would prevent you from having the vaccination. If you are booked in to a large vaccination centre, you may need to bring your booking reference number with you.
Upon arrival at your vaccination centre you will be greeted by someone to check that you do not have symptoms that would prevent you from having the vaccination. If you are booked in to a large vaccination centre, you may need to bring your booking reference number with you.
You will be asked some questions about your medical history, asked to give consent to have the vaccination and have the opportunity to ask any questions.
All places that offer vaccinations will have precautionary measures in place to prevent the spread of coronavirus. It is expected that everyone will be observing social distancing and you will be expected to wear a face covering.
The Vaccination
At present, there are two main vaccines being administered in the UK: the Pfizer vaccine and the Moderna vaccine. These vaccines have met strict standards of safety, quality and effectiveness set out by the independent Medicines and Healthcare products Regulatory Agency (MHRA). A third vaccine by Novovax is available in specialist vaccination clinics for the small proportion of people who cannot have the Pfizer or Moderna types due to contraindications.
The AstraZeneca vaccine was administered as part of the initial rollout and some booster doses but its use has now been discontinued in the UK with no further stock available.
The COVID-19 vaccine is given as an injection into your upper arm. You may be asked to wait up to 15 minutes after receiving the vaccination so that you can receive urgent care in the very rare event of an allergic reaction. If you feel unwell for any reason after receiving the vaccine, you should let the staff know.
The first dose of the COVID-19 vaccine will give you some protection from coronavirus. However, you need to have both doses of the vaccine to give you the best protection. Second doses are typically administered a minimum of 8-weeks after the first dose.
Evidence indicates high levels of protection against serious disease and death from around two weeks after the first dose. It is important to have both doses of the vaccine to give you the best protection.
When you have your first dose, you may receive a leaflet and a record card with your next appointment written on it.
Keep your record card safe and make sure you keep your next appointment to get your second dose.
If you were on immunosuppressant medications (including steroids of 10mg or more per day) when you had your first two doses, you will be recommended to have a third dose for your primary course.
Booster Doses
The level of protection provided by the COVID-19 vaccines has been shown to wane over time. Therefore, to ensure continued protection against the virus, booster doses are being offered a minimum of three months after completing your primary course. This booster dose will help extend the protection you gained from your first doses and give you longer term protection. The booster will help to reduce the risk of you needing admission to hospital due to COVID-19 infection.
The JCVI advises a preference for mRNA-type vaccines such as Moderna and Pfizer to be offered as the booster dose irrespective of which vaccine was used for your first doses. Where the Pfizer and Moderna vaccines cannot be offered (e.g. due to contraindication), vaccination with a different vaccine may be considered.
Should people with lupus have the COVID-19 vaccine?
 COVID-19 infection can be very serious, and in some cases fatal, for some people; especially older adults and those with some long-term health conditions.
COVID-19 infection can be very serious, and in some cases fatal, for some people; especially older adults and those with some long-term health conditions.
The COVID-19 vaccination will reduce the chance of you getting severe COVID-19 disease and therefore it is generally recommended that people with lupus should have the vaccine. It may take a few weeks after vaccination for your body to build up protection. No vaccine is 100% effective – some people may still get COVID-19 after having a vaccination, but this should be less severe.
Is the COVID-19 vaccine safe for people with lupus?
Any coronavirus vaccine that is approved must go through all the clinical trials and safety checks all other licensed medicines go through. The UK has some of the highest safety standards in the world.
Tens-of-thousands of people have been given a COVID-19 vaccine in clinical trials (whilst significantly larger numbers of people have now also received the vaccines – tens of millions in the UK alone). The risk of severe effects from a COVID-19 vaccine is tiny when compared with the risk of getting ill from COVID-19. It’s also important to realise that vaccine safety monitoring picks up any serious illness somebody might experience after having a vaccine, but this doesn’t mean the vaccine caused it.
You can learn more about how the COVID-19 vaccines were developed so quickly in this helpful short video from the National Institute for Health Research (NIHR).
You can read more about why vaccines are safe and important HERE.
If you would like to read about the experiences of lupus patients being administered the COVID-19 vaccines, please take a look at our online community forum, where many members have been sharing about this – https://healthunlocked.com/lupusuk
On 09/07/2021 The European Medicines Agency (EMA) issued advice that myocarditis and pericarditis (heart inflammation) can occur in very rare cases following vaccination with the Pfizer and Moderna vaccines. The symptoms of heart inflammation can include chest pain, a feeling of breathlessness and a pounding or fluttering heartbeat. Anyone with these symptoms should see a doctor. Reports of this side-effect are extremely rare, and the events are typically mild with individuals usually recovering within a short time with standard treatment and rest. Most cases are thought to be within 14 days of vaccination.
The guidance for those in doubt is to seek advice from the relevant health care practitioner.
Should I have the vaccine if I’m experiencing a lupus flare?
A lupus “flare” is when your lupus symptoms worsen and you feel ill as a result. The formal definition of a flare is: “A measurable increase in disease activity in one or more organ systems involving new or worse clinical signs and symptoms and/or lab measurements”.
If you are experiencing a lupus flare, it is recommended that you discuss the timing of your vaccination with your consultant. Your consultant will be able to recommend whether you should have your vaccine as scheduled, or to delay until the flare has passed.
What about people with a weakened immune system and those taking immunosuppressant medication(s)?
It is common for people with lupus to have a weakened immune system, either because of the disease itself, or more commonly, due to taking immunosuppressant medications.
Which medications are immunosuppressants?
Steroids can weaken your immune response, especially when taken as a daily dose of 20mg or higher.
 Immunosuppressive medications include:
Immunosuppressive medications include:
- Azathioprine
- Leflunomide
- Methotrexate
- Mycophenolate (mycophenolate mofetil or mycophenolic acid)
- Ciclosporin
- Tacrolimus
- Sirolimus
- Cyclophosphamide
- This list is not exhaustive
It does NOT include hydroxychloroquine or sulphasalazine.
Biologic medications include:
- Rituximab
- Belimumab
- Anifrolumab
- This list is not exhaustive
What effect will immunosuppression have on the vaccine?
All COVID-19 vaccines currently being administered in the UK are safe for people with weakened or suppressed immune systems.
Current evidence suggests that vaccine effectiveness is reduced for those who are immunosuppressed. For some, though not all, immunosuppressed people, this is improved by further doses. You can find more information in the links below.
- This webinar from the British Society of Immunology on how effective vaccines are in people with weakened immune systems.
- This information from the UK Covid Vaccine Research Hub for people with weakened immune systems, which includes links to ongoing and published research studies.
- This review of studies investigating vaccine response in immunocompromised people (from January 2022; written for scientific audience)

PHE has suggested that measurement of antibody levels post-vaccination appears to generally indicate the likelihood of vaccine protection against infection. However, they also warned that this does not fully assess the likely level of protection, as antibody levels are not the only factor. The reasons antibody tests may not show the full picture include:
- The presence of antibodies almost certainly indicates protection from severe disease, but doesn’t mean protection from infection.
- Antibody levels almost certainly reduce over time, so tests only capture levels at the time of testing, not future levels of protection.
- Other aspects of the immune system are also involved in the response to infection, and particularly important are a type of blood cell called T-cells. The level of T-cell response is not easily assessed and is not measured at all by antibody tests. However, it’s very rare for only your T-cells to respond to COVID-19 infection, without some involvement of antibodies.
Antibody testing is therefore the most accessible way of indicating vaccine protection against infection at the time of testing, but does not fully assess the potential immune response.
Should I temporarily stop my medication when I have the vaccine?
The VROOM (Vaccine Response On Off Methotrexate) Study investigated the effect of pausing methotrexate on COVID-19 vaccine efficacy. You can read about the study findings HERE.
The clinical trial found that people who paused their methotrexate for two weeks after a COVID-19 booster dose would have more than twice as much antibody against COVID-19 spike-protein after the vaccination compared to those that continued treatment. People who paused their methotrexate were also more likely to have a flare of their disease, most of which were self-managed.
The British Society for Rheumatology (BSR) have updated their guidance about COVID-19 vaccination to reflect the findings from the VROOM Study. You can see their full guidance HERE.
If you are taking methotrexate, talk to your hospital team or GP before your next COVID-19 booster dose. They will advise you on the best course of action for you, taking your preference, condition, and how well your disease is controlled into account.
At present there is insufficient evidence about the effect of pausing other immunosuppressive medications for COVID-19 vaccination.
Please note that you should never adjust your immunosuppressant or steroid treatment unless advised by your doctor.
Advice may vary on a case-by-case basis to maximise the chance of effect from the vaccine whilst managing lupus disease activity. It is important to discuss the timing of your vaccine with your consultant if you are due to have an infusion of rituximab.
What should I do if I am due to start an immunosuppressant medication soon?
Some people may be due to start on a new immunosuppressant medication, especially if they are newly diagnosed, experiencing a flare or unable to tolerate other treatments.
The JCVI Green Book Chapter 14a was updated on 21st January 2021. The updated guidance recommends that any patients who are due to receive planned immunosuppressive therapy should be considered for vaccination (ideally two weeks) before commencing that treatment. Where possible, it is also advised to complete doses of the vaccine before commencing immunosuppression – this could involve offering subsequent vaccine doses at the recommended minimum (three or four weeks). Any decision to defer immunosuppressive therapy or to delay possible benefit from vaccination needs to be carefully considered.
Will I be offered a third “primary” vaccine dose?
 People with “severely weakened immune systems” should have a third “primary” COVID-19 vaccine dose. The recommendation is for third primary doses to be offered to people over 12 who were severely immunosuppressed at the time of their first or second dose. These people may not mount a full response to vaccination and therefore may be less protected than the wider population. These third doses should be considered part of the primary dosing and are separate to the “booster” programme.
People with “severely weakened immune systems” should have a third “primary” COVID-19 vaccine dose. The recommendation is for third primary doses to be offered to people over 12 who were severely immunosuppressed at the time of their first or second dose. These people may not mount a full response to vaccination and therefore may be less protected than the wider population. These third doses should be considered part of the primary dosing and are separate to the “booster” programme.
Most individuals whose immunosuppression commenced at least 2 weeks after the second dose of vaccination do not require a third dose.
What counts as “severely immunosuppressed”?
Updated guidance from the British Society for Rheumatology (BSR) states, “Based on the JCVI recommendations, nearly all rheumatology patients (aside from those solely on hydroxychloroquine or sulfasalazine), should receive a third dose.” You can access the BSR guidance in full, HERE.
The JCVI have issued a report (HERE) indicating which people they recommend should be considered eligible for a third vaccine dose. This includes:
1. Individuals with primary or acquired immunodeficiency at the time of vaccination.
This could be due to conditions including leukaemias, HIV/AIDS, stem cell transplants or persistent agammaglobulinaemia. A more detailed list can be found in the report.
2. Individuals on immunosuppressive or immunomodulating therapy at the time of vaccination.
This includes:
– Those who were receiving or had received immunosuppressive therapy for a solid organ transplant in the previous 6 months.
– Those who were receiving or had received in the past three months a targeted therapy for autoimmune disease such as biologic immune modulators including B-cell targeted therapies (i.e. belimumab and rituximab) – in the case of rituximab the recipient would be considered immunosuppressed for a 6 month period.
– Those who were receiving or had received in the previous 6 months immunosuppressive chemotherapy or radiotherapy for any indication.
3. Individuals with chronic immune-mediated inflammatory disease (such as lupus) who were receiving or had received immunosuppressive therapy prior to vaccination:
This includes:
– High dose corticosteroids (equivalent to 20mg or more of prednisolone per day) for more than 10 days in the previous month.
– Long-term moderate dose corticosteroids (equivalent to 10mg or more of prednisolone per day for more than 4 weeks) in the previous 3 months.
– Non-biological immunosuppressant treatments such as:
- Methotrexate – more than 20mg per week (oral and subcutaneous)
- Azathioprine – more than 3mg/kg/day
- Mycophenolate – more than 1g/day in the previous 3 months
- certain combination therapies at individual doses lower than above, including those on 5mg or more of prednisolone per day in combination with other immunosuppressants (other than hydroxychloroquine or sulfasalazine) and those receiving methotrexate (any dose) with leflunomide in the previous 3 months.
4. Individuals who had received high dose steroids (equivalent to more than 40mg prednisolone per day for more than a week) for any reason in the month before vaccination.
Which vaccine will I be offered for the third dose?
For those aged 18 years and over, the JCVI advises a preference for mRNA vaccines (Moderna and Pfizer) for the third dose. For those aged 12 to 17 years the Pfizer vaccine remains the preferred choice.
What is the risk of side-effects from a third dose?
Data from Pfizer has shown that the third dose shows similar side-effects and adverse reactions compared with those experienced with the second dose. Side-effects have not been shown to get worse. Third dose rollouts have already taken place in Israel and the USA, where this data was collected.
When will I be offered a third dose?
The third primary dose should ideally be given at least 8 weeks after the second dose. In some cases, special attention needs to be given to current or planned immunosuppressive therapies to calculate the most appropriate timing to generate the best immune response.
If you live in England, are eligible for a third primary dose, and have not yet received an invitation, you can now book online – HERE.
Information about the third primary dose for people living in Scotland can be found HERE. Information for people living in Wales can be found HERE.
How do I know the third dose will offer additional protection?
Research has suggested that a third dose can improve protection for some, but not all, immunocompromised people (for example this research study on third doses in people with inflammatory bowel disease who are immunosuppressed, and this one on people with cancer). Findings from the OCTAVE study indicated that people treated with rituximab may be at particular risk of not generating a measurable immune response. However, a third dose is offered to all immunosuppressed people on the assumption that is it is unlikely to be harmful but may benefit.
If I have a third dose, will I also need a booster jab?
Yes. The third dose is counted as part of your primary course. Booster doses are recommended with the first being offered a minimum of three months after receiving your third primary dose.
Do I need a booster dose?
 It is advised that all people with lupus should have booster doses, if invited. Like some other vaccines, levels of protection appear to reduce over time. Booster doses will help extend the protection you gained from your primary course and give you longer term protection. The booster will help to reduce the risk of you needing admission to hospital due to COVID-19 infection.
It is advised that all people with lupus should have booster doses, if invited. Like some other vaccines, levels of protection appear to reduce over time. Booster doses will help extend the protection you gained from your primary course and give you longer term protection. The booster will help to reduce the risk of you needing admission to hospital due to COVID-19 infection.
When will I be invited for a booster dose?
You are eligible for your first booster at least three months after your last primary dose. The booster dose should help to extend your protection. You will be able to book this when you have been invited (see section below on who is eligible for the 2023 booster).
England – BOOK ONLINE
Scotland – BOOK ONLINE
Wales – CHECK YOUR LOCAL HEALTH BOARD
Northern Ireland – BOOK ONLINE
 Information on the spring 2024 booster dose programme
Information on the spring 2024 booster dose programme- All adults aged 75 years and over
- Residents in a care home for older adults
- Individuals aged 6 months and over who are immunosuppressed.
(“Immunosuppressed” is defined in tables 3 and 4 of the COVID-19 immunisation Green Book HERE.)
When will invitations for the spring 2024 booster be sent?
The NHS has not yet released any details about when the spring 2024 vaccines will begin. We will update this page when we have more information.
Which vaccines will be offered in spring 2024?
The JCVI have not yet recommended which vaccines should be used. We will update this page when they do.
Which vaccines were offered in spring & autumn 2023?
- a bivalent version of Modern’s COVID-19 vaccine
- a bivalent version of Pfizer’s COVID-19 vaccine
- Sanofi/GSK‘s COVID-19 vaccine (also called VidPrevtyn Beta)
- Novavax COVID-19 vaccine (only when people are unable to receive other available vaccines).
What if I cannot have the Moderna or Pfizer vaccine?
Some people may not be able to have the Moderna or Pfizer vaccine because of a contraindication such as severe allergy or previous adverse effect. Previously, these people may have been offered the AstraZeneca vaccine but this vaccine is no longer being used in the UK, with all stocks exhausted. Those people who cannot have a Moderna or Pfizer vaccine may be offered a Novovax vaccine through a specialist clinic. You can read more about this vaccine HERE. If a recent clinical assessment confirms that you cannot have one of the common vaccine types, your GP practice or vaccination centre should refer you to a specialist clinic – more information can be found HERE.
I had an adverse reaction to a previous dose of the vaccine, should I have the booster?
If you experienced significant side-effects or an adverse reaction, such as a lupus flare, from any previous dose of the COVID-19 vaccine, you may be advised to avoid or delay further vaccination. You should discuss this with your doctor or specialist.
I am on strong immunosuppressive therapy, is there any point in having the booster?
There is a small proportion of people who may have had no measurable antibody response to any of the previous COVID-19 vaccinations. This is of particular concern for kidney transplant recipients and people treated with anti-CD20 therapies such as rituximab and obinutuzumab.
Despite the poor measurable antibody response, it is still recommended that people on strong immunosuppressant treatments have the autumn booster. Immune protection is not solely based on antibody protection and the potential benefit of the booster dose is considered to outweigh the possible risks.
I’m also due to have the annual flu vaccine. Can I have both vaccines at the same time?
It is generally recommended that people with lupus should have the annual flu vaccine. You can read more about this in our article HERE. Administration of these vaccines may vary regionally, with some administering them at the same time (in different arms) and some administering them separately. There is no concern about the safety or efficacy of both the COVID-19 and flu vaccines being administered at the same time. If you would prefer to have the vaccines administered separately, please notify vaccination centre staff on arrival so that a separate flu vaccine appointment can be booked.
 Children are generally at low risk of developing severe COVID-19, but children with underlying conditions have a higher risk of being admitted to hospital.
Children are generally at low risk of developing severe COVID-19, but children with underlying conditions have a higher risk of being admitted to hospital. 
Approximately 1-in-5 people with SLE will also have an overlapping diagnosis of antiphospholipid syndrome (learn more about this HERE). As a result, some lupus patients will be taking anticoagulation medication (blood thinners), such as aspirin or warfarin.
Thrombosis UK have provided helpful guidance for anyone taking this type of medication:
Individuals receiving direct oral anticoagulant (apixaban, dabigatran, edoxaban & rivaroxaban) or warfarin in therapeutic INR range, on full dose heparin or fondaparinux injections or antiplatelet medications such as aspirin and clopidogrel can all receive the COVID-19 vaccination.
Please beware of the following issues:
- There is a risk of bruising at the injection site, but we do not anticipate any serious effects related to anticoagulation.
- We suggest that after the injection, prolonged pressure (at least 5 minutes) should be applied to the injection site to reduce bruising.
- Patients on warfarin with supra-therapeutic INR should wait until their INR is <4.0.
- We encourage patients to have vaccinations and they should not be avoided on the basis of being on anticoagulation treatment.
Who SHOULD NOT have the COVID-19 vaccine?
According to the JCVI Green Book there are very few individuals who cannot receive the vaccine. Following careful monitoring of the vaccine rollout, they have issued specific guidance for those with allergies and those who have had an allergic reaction to a previous dose of the vaccine. Their advice is as follows:
For those with a history of allergic reaction to any component of a COVID-19 vaccine:
- People who have a history of systemic allergic reaction to a component of the vaccine should be referred to a specialist. The person may be offered an alternative vaccine which does not contain their allergens, or have a vaccine administered under medical supervision at a hospital
- The full contents of the Pfizer vaccine can be found here. The full contents of the Moderna vaccine can be found here. The full contents of the Novovax vaccine can be found here. The full contents of the Sanofi vaccine (VidPrevtyn Beta) can be found here.
For those with a confirmed anaphylaxis reaction to a previous COVID-19 vaccine:
- An alternative vaccine may be offered.
- The same vaccine may be offered under close medical supervision
For those with a non-anaphylaxis reaction to a previous COVID-19 vaccine:
- Where there were no signs of anaphylaxis and the symptoms rapidly resolved, then the same vaccine may be offered as evidence suggests people are able to tolerate a second dose.
For those with a history of immediate anaphylaxis to multiple different drug classes:
- These individuals should not be offered the Moderna or Pfizer BioNTech vaccines, except on the expert advice of an allergist or where the person has previously tolerated a dose of the same vaccine.
- An alternative vaccine may be offered with close monitoring.
For those with a history of allergic reactions (including anaphylaxis) to food, medicines, insects, etc:
- People with a history of reactions, but not to an ingredient in one of the COVID-19 vaccines, to a previous COVID-19 vaccine, or to multiple classes of drugs, can receive the vaccine in any setting.
Other common concerns:
- There is no known risk of having the vaccine while pregnant or breastfeeding.
- The Sanofi vaccine (VidPrevtyn Beta) contains an oil derived from fish. None of the other currently available vaccines contain any animal products or egg.
- The vaccines are suitable for people of all faiths.

You should also wait to have your vaccination (first or second dose) if:
- you are currently unwell with a fever.
- you have had another vaccine in the 7 days before your planned appointment.
- you have COVID-19 test or suspect you may have COVID-19 (to avoid infecting others at your vaccination venue)
- you have prolonged COVID-19 symptoms and have recently deteriorated or are seriously debilitated.
Is the COVID-19 vaccine effective?
 The first dose of the COVID-19 vaccine will most likely give you good protection against serious disease from coronavirus. However, you need to have both doses of the vaccine to give you the best protection. It also seems important to have both doses of the vaccine for longer-term protection from the virus.
The first dose of the COVID-19 vaccine will most likely give you good protection against serious disease from coronavirus. However, you need to have both doses of the vaccine to give you the best protection. It also seems important to have both doses of the vaccine for longer-term protection from the virus.
Evidence indicates high levels of protection against serious disease and death.
A study in the USA indicated that the COVID-19 vaccine booster reduced risk of infection in people with lupus. You can read the study HERE.
Like all medicines, no vaccine is completely effective – some people may still get COVID-19 despite having a vaccination, but this should be less severe.
This means it is important to continue following social distancing guidance.
Will the vaccine stop someone from spreading the virus?
The COVID-19 vaccine will not entirely remove the risk of contracting or passing on the virus. It is therefore very important to continue following the guidance in your local area to protect those around you.
Does the COVID-19 vaccine have side-effects?
Like all medicines, vaccines can cause side-effects. Most of these are mild and short-term, and not everyone gets them.
 Very common side-effects include:
Very common side-effects include:
- having a painful, heavy feeling and tenderness in the arm where you had your injection. This tends to be worst around 1-2 days after the vaccine.
- feeling tired.
- headache.
- general aches, or mild flu-like symptoms.
It is not possible to contract COVID-19 infection from having the vaccination.
Although feeling feverish is not uncommon for 2 to 3 days, a high temperature is unusual and may indicate you have COVID-19 or another infection. You can rest and if you would normally be able to safely take painkillers, such as a normal dose of paracetamol (follow the advice in the packaging), then you can do so to help you feel better.
Symptoms following vaccination normally last less than a week. If your symptoms seem to get worse or if you are concerned, call NHS 111. Even if you do have symptoms after the first dose, you will most likely be recommended to have the second dose. Although you may get some protection from the first dose, having the second dose will give you the best protection against the virus. However, this advice may depend on the severity of your side-effects and you should discuss this with your consultant if unsure.
Some research has been done to investigate how well tolerated the COVID-19 vaccines are by people with lupus. The international VACOLUP study (HERE) found that 45% of patients reported a side-effect after the first vaccine dose and 53% after the second vaccine dose. 3% of patients reported a medically confirmed SLE flare, typically with musculoskeletal symptoms and/or fatigue. Having a flare during the past year before vaccination was associated with an increased risk of SLE flare after COVID-19 vaccination.
The EMA & MHRA are also continuing to investigate myocarditis and pericarditis (heart inflammation) which can occur in very rare cases following vaccination with the Pfizer and Moderna vaccines. The symptoms of heart inflammation can include chest pain, a feeling of breathlessness and a pounding or fluttering heartbeat. Anyone with these symptoms should see a doctor. Reports of this side-effect are extremely rare, and the events are typically mild with individuals usually recovering within a short time with standard treatment and rest. Most cases are thought to be within 14 days of vaccination.
If you do seek advice from a doctor or nurse, make sure you tell them about your vaccination (show them the vaccination card if possible) so that they can assess you properly.
You can also report suspected side effects to vaccines and medicines online through the Yellow Card scheme.
It is very rare for anyone to have a serious reaction to the vaccine (anaphylaxis). If this does happen, it usually happens within minutes. Staff giving the vaccine are trained to deal with allergic reactions and treat them immediately.
If you would like to read about the experiences of lupus patients being administered the COVID-19 vaccines, please take a look at our online community forum, where many members have been sharing about this – https://healthunlocked.com/lupusuk
Which COVID-19 vaccine will I be given?
At present, the main vaccines being used in the UK are the Pfizer vaccine and the Moderna vaccine.
If these types of vaccines are contraindicated for you as an individual, you be offered a Novovax vaccine through a specialist clinic.
Further Reading
NHS – Coronavirus (COVID-19) vaccine
NHS – Coronavirus Vaccination – What happens at your appointment
UK Government – COVID-19 vaccination: Guide for older adults
ARMA – Principles for COVID-19 Vaccination in Musculoskeletal and Rheumatology for Clinicians
Public Health England – COVID-19 Vaccination Programme – Information for Healthcare Practitioners
British Society for Rheumatology – COVID-19 Guidance
The Green Book – COVID-19 Chapter
Department of Health & Social Care – UK COVID-19 vaccines delivery plan
We are extremely grateful to Prof David Jayne (Director of the Vasculitis and Lupus Service, Addenbrooke’s Hospital), Prof Chris Edwards (Consultant Rheumatologist, University Hospital Southampton) and Dr Rona Smith (Clinical Lecturer in Nephrology and Experimental Medicine, Addenbrooke’s Hospital) for reviewing the content of this article.


 ©2024 LUPUS UK (Registered charity no. 1200671)
©2024 LUPUS UK (Registered charity no. 1200671)